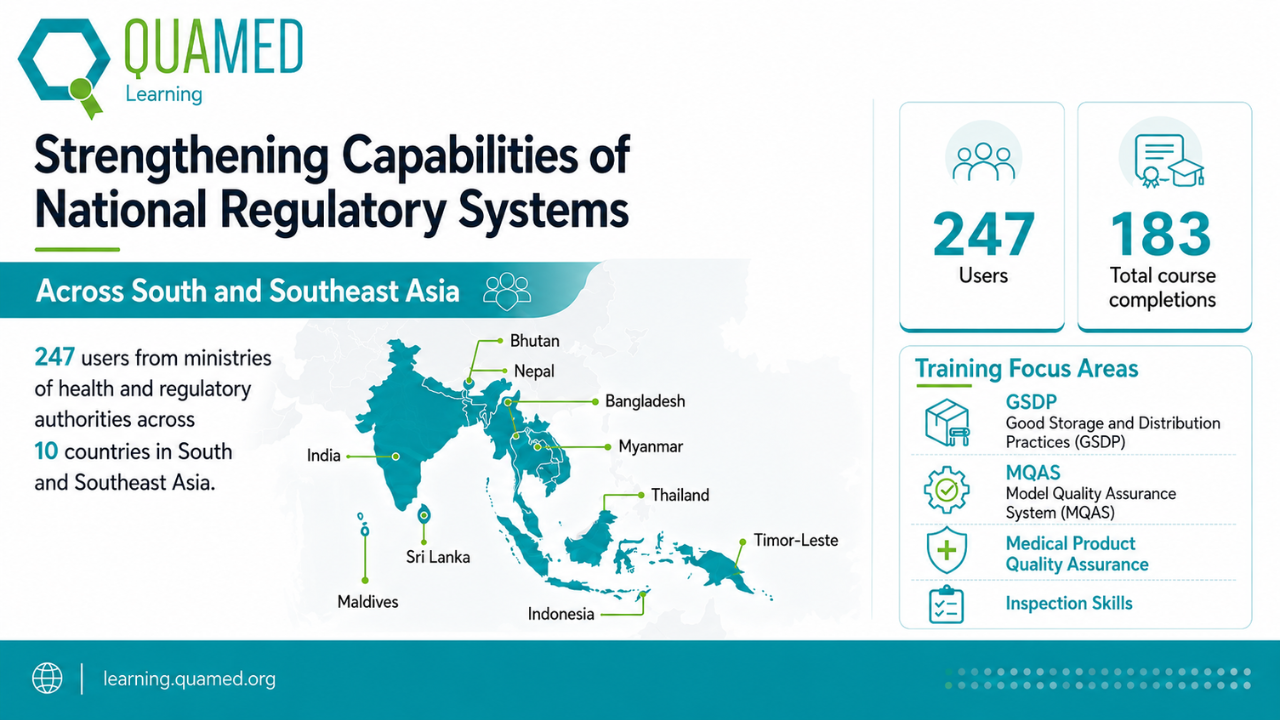
QUAMED Learning is supporting professionals from ministries of health and regulatory authorities across Bangladesh, Bhutan, India, Indonesia, Maldives, Myanmar, Nepal, Sri Lanka, Thailand, and Timor-Leste. Across these countries, 247 users are engaging with the platform, with 183 course completions recorded so far across QUAMED Learning courses. This engagement reflects the growing commitment of public-sector professionals ...